A Quick Guide to Drug Pharmacokinetics and Pharmacodynamics
We all know the general idea about how medications work, right? Drugs that save lives, control diseases, and even prevent or delay certain ones from developing in the first place. In addition, not only do they come in various forms, but they’re also taken into the body in ways other than the mouth. It’s fascinating to see the evolution of drugs over the years and the invention of new ones. What’s even more fascinating is the way that these magic bullets work in the body. Thanks to the brilliant teamwork of pharmacokinetics and pharmacodynamics, drugs are able to do what they do. Let’s navigate these concepts together.
Pharmacokinetics: The Body’s Effect on the Drug
Pharmacokinetics has to do with what the body does to a drug and involves 4 processes: absorption, metabolism, distribution, and excretion.1
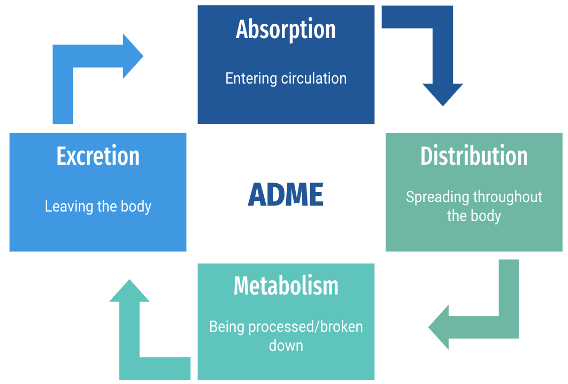
Step 1: Absorption
- Once a drug is administered, (via the oral, intravenous, intramuscular, subcutaneous etc. routes) it enters systemic circulation. The method of administration has its own characteristics of absorption which affect the speed and amount of drug that reaches the desired location of effect.
Step 2: Distribution
- The drug travels through the blood to be dispersed throughout the tissues. How well it gets distributed depends on the biochemical properties of the drug and the physiology of the person. The end goal is to achieve effective drug concentration at the desired destination.
Step 3: Metabolism
- Most drugs pass through to the liver which is the main site of drug metabolism. Active drugs are either converted to inactive forms that are more hydrophilic to move on to renal clearance, or in the case of a prodrug, it is subsequently converted into active metabolites.
Step 4: Excretion
- The drug reaches the final step in its journey through the body where it can now be removed. The kidneys usually perform this major function with the drug excreted via urine. Other forms of excretion are bile, sweat, or stool.
Pharmacodynamics: The Drug’s Effect on the Body
Pharmacodynamics has to do with what the drug does to the body and involves the drug’s characteristics and physiological effects. Drugs produce effects by interacting with the body either through direct or indirect means. Direct effects result from the direct interaction of the drug with a receptor or enzyme that produces the effect. Indirect effects result from an interaction of the drug with a receptor or protein somewhere else that produces the desired effect. The interaction with specific receptors, enzymes, or molecules is what leads to desired therapeutic effects or undesired side effects.
The Balancing Act
It’s not enough to select the right drug. Finding the right dose is also key. It is essentially a balancing act. Too much of a drug can be toxic while too little may not produce the intended effect. Considering factors such as the drug’s physical and chemical properties, the characteristics of the patient it’s being given to (because no two individuals are the same), and the correct dose and dosage form of the drug are all paramount to achieving balance and the desired result. The teamwork between pharmacokinetics and pharmacodynamics is a captivating spectacle that simply could not work without each working alongside the other.

Midrara Kashmari
RxPharmacist Team
References:
- Grogan S. Pharmacokinetics. PubMed. Published 2020. https://www.ncbi.nlm.nih.gov/books/NBK557744/.
- Marino M, Zito PM. Pharmacodynamics. PubMed. Published 2020. https://www.ncbi.nlm.nih.gov/books/NBK507791/.
A Quick Guide to Drug Pharmacokinetics and Pharmacodynamics Read More »